- Blog
- 4k dirt 3 background
- Fable 2 download for pc
- Reason 11 crack download
- Screenshot of primary monity in windows 7
- See finance android
- Corpse party seiko art
- Runescape classic auto caster
- Mindview behavioral health
- Goodnotes free templates
- Amazing audio player will not save
- Euro truck simulator 2 free download
- F150 lightning price
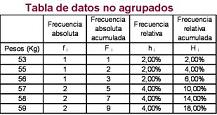
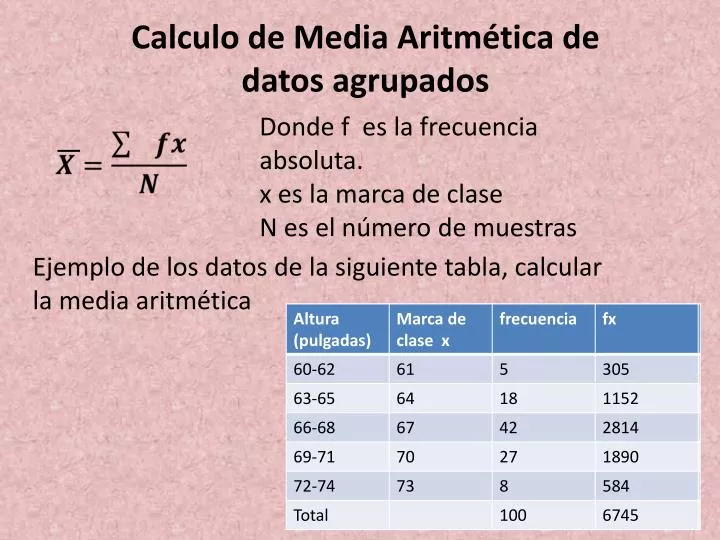
- Dato agrupados
The work that we present is part of a multidisciplinary project to recover and disseminate information about the Reggio Calabria’s (southern Italy) artistic, archaeological and cultural heritage. We underline the importance of an integrated visualization from an archaeological and architectural perspective to obtain understanding of the structure with the integration of the two models with different data. However, if this is not possible, technologies that use 3D Virtual Reality help to provide a small knowledge base to those who cannot use the museum. In fact, visiting a cultural heritage site in person allows one to receive much more information on finds and structures. This does not mean, of course, that with the mere creation of a 3D model (and allowing it to be viewed in 3D), the public automatically obtains more information about heritage. One of the peculiarities of these integration techniques is the possibility of promoting the dissemination of knowledge through virtual reality, augmented reality and mixed reality, given the widespread use of mobile devices. The possibility of integrating three-dimensional models from different acquisition systems (laser scanner, UAV, reflex and Georadar) is even more exciting. Three-dimensional digital acquisition techniques can be useful in archaeology because they make a further technological contribution to the visualization of finds and structures.

Most of the interacting amino acid residues of the receptor fall within the RBD (438–506). The binding affinities of anti-spike drugs varied among different variants.

On the other hand, fenofibrate showed the least binding affinity towards the RBD of the S1 protein of all eleven variants. Ceftazidime was the second-best drug in terms of binding affinity towards the S1 RBD of the investigated variants. Cefoperazone was found to possess the highest binding energy to the RBD of the S1 protein of all the eleven variants. The docking results revealed that all tested drugs possess moderate to high binding energies to the receptor-binding domain (RBD) of the S1 protein for all different variants. The S1 protein subunit from eleven different variants, including the latest highly contiguous omicron variant, were used as targets for the docking study. Seven FDA approved drugs, namely, camostat, nafamostat mesylate, fenofibrate, umifenovir, nelfinavir, cefoperazone and ceftazidime, were selected based on their reported in vitro and clinical activities against SARA-CoV-2. The current study intended to assess the ability of different variants of interest (VOIs) and variants of concern (VOCs) of SARS-CoV-2 for their affinities of binding to different repurposed drugs.
Although many mutations have an insignificant impact on the virus properties, mutations in the surface protein, especially those in the receptor-binding domain, may lead to immune or vaccine escape variants, or altered binding activities to both the cell receptor and the drugs targeting such a protein. Like most of the RNA viruses, SARS-CoV-2 continuously mutates.